Ready-To-Glow Secreted Luciferase Reporter Assay
Ready-To-Glow Secreted Luciferase Reporter Assay
Our Ready-To-Glow Secreted Luciferase Reporter Assay is a complete system designed for use in a transcription reporter system based on luciferase activity in the supernatant of transfected cells. In the presence of secreted luciferase in the sample, a nonluminescing substrate is converted to a luminescent compound. The intensity of bioluminescence is directly proportional to the amount of luciferase activity. The kit, which includes a substrate buffer, reaction buffer, and a lyophilized secreted luciferase substrate, is available in three reaction sizes. Our Ready-To-Glow Secreted Luciferase Reporter Assay has been customized to work optimally with our Secreted Luciferase Vector Kits.
Overview
- Secreted luciferase assay protocol eliminates cell lysis
- 2–4 fold higher signal than firefly or Renilla luciferase
- Single transfection allows multiple time points from same cell
- Dual, live-cell assay includes SEAP as well as secreted luciferase:
monitor two promoters, or use one reporter as a normalization control
Applications
- Homogeneous assay protocols
- Time-course studies
- Test multiple compounds
- Suitable for downstream experiments with the same cells
- Use the dual assay (which includes secreted alkaline phosphatase, as well as a secreted luciferase assay) to monitor two promoters
- Use the dual assay to monitor one promoter with a built-in normalization control
Limit of detection for recombinant Metridia luciferase protein

Limit of detection for recombinant Metridia luciferase protein. The indicated amounts of recombinant Metridia luciferase (fg/well) were spiked into DMEM containing 10% FBS. Metridia luciferase activity was measured in a 96-well format on the SpectraMax L. The limit of detection was determined to be 2 fg/well (40 fg/ml).
High signal intensity and stability using secreted Metridia luciferase

High signal intensity and stability using secreted Metridia luciferase. CHO cells were plated into 96-well plates and transiently transfected with CMV-driven constructs encoding non-secreted firefly luciferase, non-secreted Renilla luciferase, and secreted Metridia luciferase. 24 hr after transfection, luciferase activity in equivalent samples was analyzed by addition of the recommended substrate. The signal was measured at different timepoints over a period of 45 min. neg = negative control.
Monitoring promoter activation using the sequence-optimized secreted Metridia luciferase reporter

Monitoring promoter activation using the sequence-optimized secreted Metridia luciferase reporter. HeLa cells were transiently transfected with a vector construct containing the NFkB response element driving the expression of sequence-optimized secreted Metridia luciferase. 24 hr after transfection, the media was removed and replaced by media with or without TNF-α (100 ng/ml) to activate the NFkB signal transduction pathway. Six hr after addition of TNF-α, samples of the media were removed and analyzed for Metridia luciferase activity. The fold induction was calculated for different time points following the addition of substrate.
Flow chart of the Ready-To-Glow Secreted Luciferase Reporter System
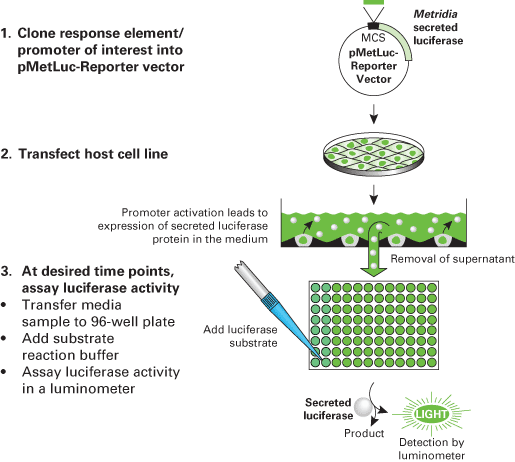
Flow chart of the Ready-To-Glow Secreted Luciferase Reporter System.
Flow chart of the Ready-To-Glow Secreted Luciferase Reporter System

Flow chart of the Ready-To-Glow Secreted Luciferase Reporter System.
Use of secreted Metridia luciferase in a high-throughput screening (HTS) application

Use of secreted Metridia luciferase in a high-throughput screening (HTS) application. The figure shows a screen shot from a CCD camera of a 1,536-well plate containing stable CHO cells transfected with a forskolin-responsive Metridia luciferase gene (pASM-Lu164). 300 cells/well were plated in a 1,536-well microtiter plate. Cells were incubated with increasing concentrations of forskolin and the plate was incubated for 4 hr at 37° C. Metridia substrate was added in increasing concentrations and the plate was visualized using a CCD camera system (integration time: 60 sec). (Courtesy of Bayer Health Care, Germany). This pseudocolor image reflects luminescence intensity. Yellow regions are the brightest, while black regions are the least bright.